About ZYLiQ
ZYLIQ is a platform designed to help regulatory writers to automate the creation of scientific documents using its own unique blend of Gen-AI.
ZYLIQ is the leader in Closed-Loop Gen-AI for regulatory writing.This means that our AI models are custom-fit for your regulatory writing. It also means that our AI will never be exposed or influenced by any other regulatory documents but your own.
Unlike other Regulatory Writing AI tools, ZYLIQ is a one-process solution, so you don't have to spend time with numerous small-batches.This results in a 70% reduction in time and effort of overall regulatory writing. You can experience this firsthand with our free 2-week Pilot.
Here are some of our customers moving 70% faster with ZYLIQ
Benefits
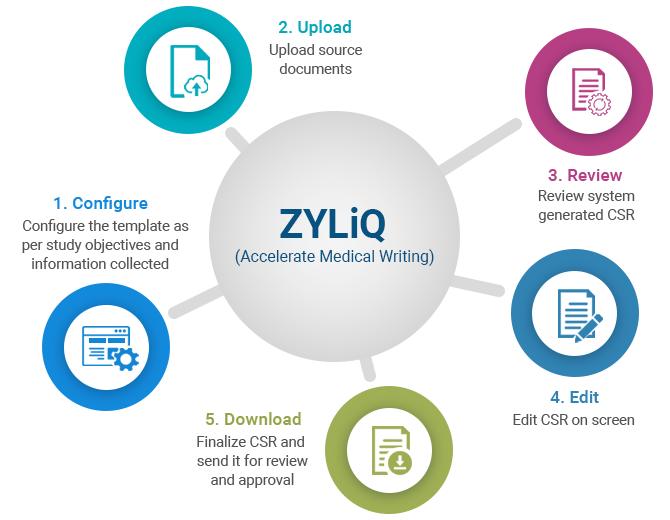
ZYLIQ uses a closed-loop, proprietary blend of Gen-AI/ML to generate the first-draft Clinical Study Reports (CSRs) using only the information from Protocol, SAP, and in-text tables.
It can also interpret certain sections of the study results, accelerating the overall CSR process by 70%. This allows medical writers to avoid the “drudgery” copy/paste to quickly focus on interpreting the study results.
Additionally, the system can be configured to integrate into workflows based on the sponsor's needs.
AI-Powered Application for
Automating Clinical Study Report (CSR) Authoring
Sponsor template
configuration
Sponsor workflow
integration
Lean Writing for
sections
Interpretation for In-text
and safety narratives
Automatic Tense
Conversion
Converting Post-text to
In-text tables
Produce Reports
(Traceability/Audit log/Version history)
Multi-authoring using MS
word like editor


